 
The case of dichlorocarbene, with a difference in AH of 15 kcal/mol), is particularly disturbing and has been analysed in some detail.134 Notably, the PA of CCl2 from an earlier bracketing experiment126 was closer to the enthalpy-derived PA. The proton affinities obtained by ICR bracketing125 (Table 6) are consistently lower than those derived from enthalpies of formation (Table 7). 8 and 9 have both been applied to halocarbenes. Thermonator 3.These gases have lower thermal stabilities than hydrogen sulphide as expected from their enthalpies of formation Table 10.2) and they are consequently more powerful reducing agents than hydrogen sulphide. Related software: EES (Engineering Equation Solver), OpenCalphad, Thermo-Calc, ProPhyPlus, BibPhy, DiagSim, ThermoSoft and many more. Languages: Write me to improve the translation into your mother tongue. Substances: Water, Air, Ammonia, Carbon Dioxide, Nitrogen, Oxygen, Alkanes (Butane, Ethane, Heptane, Hexane, Isopentane, Methane, Octane, Pentane, Propane), Alkenes (Ethylene, Propylene), Refrigerants (R11, R12, R13, R14, R22, R23, R114, R123, R134a, RC318, R500, R502, Propyl alcohol), Ideal gases (Water, Air, Ammonia, Carbon monoxide, Carbon dioxide, Methane, Acetylene, Ethylene, Hydrogen, Nitrogen (N), Nitrogen monoxide, Oxygen, Hydroxide (OH), Chlorine, Fluorine, Hydrogen bromide, Hydrogen chloride, Hydrogen iodide, Hydrogen sulfide, Sulfur dioxide), Noble gases (Helium, Neon, Argon, Krypton, Xenon, Radon). Units: Pressure (bar, at, atm, kPa, MPa, psia), Temperature (K, C, R, F), Volume (m3, ltr, ft3, gal), Mass (mol, kmol, kg, lbm), Energy (J, kJ, kc, kWh, BTu), Power (W, kW, hp, hp (UK), BTus, BTUh). 
Cycle properties: Thermal performance, COP heating, COP cooling, and all process properties. Process properties: Heat, Work, Expansion work, Technical work, Flow work, and Increments of the main state properties. 
State properties: Pressure, Temperature, Volume, Internal energy, Enthalpy, Entropy, Exergy, Pressure, Specific heat, Vapor title, and Saturated properties. Information on substances: Formula, molecular weight, critical point, specific heat, etc. Educational Aids: Quick reminders on thermodynamic concepts and formulas. Interactive diagrams (Pressure-Volume, Temperature-Entropy, Enthalpy-Entropy, Mollier, etc) with linear and logarithmic scales, automatic zoom and draggable rulers. First Principle (∆U = Q - W, ∆U = Q + W) according to different bibliographies. Reference State to adjust the results to different books or bibliographic sources: Cengel - Boles, Moran - Shapiro, Sonntag - Borgnakke - Van Wylen, Rogers - Mayhew, Bejan, Nag, Khurmi, etc. Ambient State to calculate relative pressure and exergies. Mass of Control or Volume of Control (closed system or open system) to calculate the work of the processes according to the exercise. Calculation of heat exchangers: open, closed and mixed. Mass and energy balances according to the first principle. Automatic calculation of predefined cycles: Carnot, Otto, Diesel, Dual-Mixed, Stirling, Joule-Brayton, Regenerative exchanger, Ideal power Rankine, Basic cooling Rankine, and Rankine with open or closed heat exchangers. Calculation of isobar, isothermal, isochoric, adiabatic (isentropic) and isenthalpic processes. Calculation of properties of states, processes and thermodynamic cycles of free design. Explanations of the main concepts, equations and thermodynamic laws. 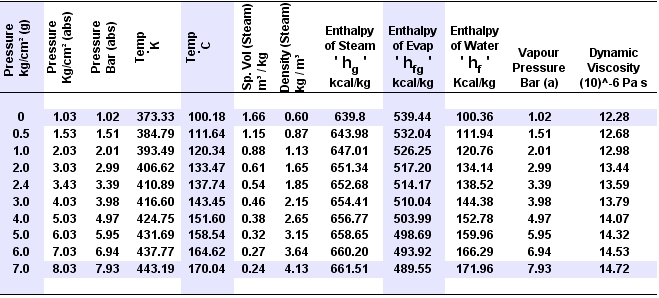
Quick and reliable calculations of steam tables of water, air, ideal gases, refrigerants and other substances.App to solve problems and exercises from Thermodynamics Engineering books. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
